Overview
The Neuroscience Program at IPI aims to empower molecular neuroscientists by developing antibody toolkits for entire receptor-ligand families involved in axon guidance, synapse formation and glial cell phenotyping.
We work with communities to validate antibody panels, identify new leads and test antibody formats for applications in fluorescent microscopy and immunohistochemistry. Our team investigates ligand-receptor interactions and leverages machine learning and AI methods to screen for application-specific and functional antibodies, working toward wide adaptation in the molecular neuroscience community.

Current activities
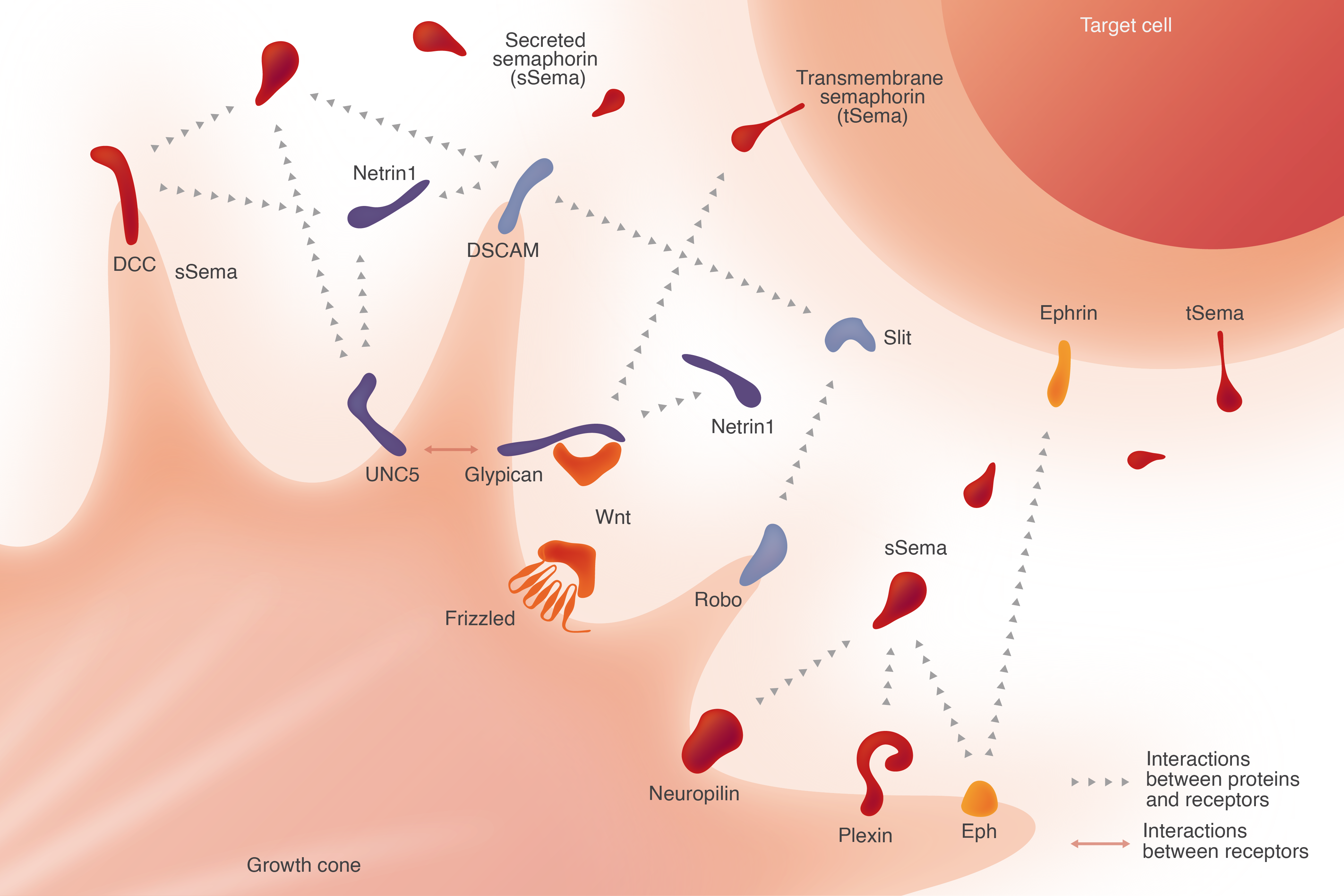
Neurons specialize in storing and transferring information through a dense network of receptors and ligands. The full breadth of this network has only recently come into view (Zhu et al., 2025), and an understanding of the relationship between the function of individual neurons and the receptors decorating the cell membrane requires protein tools that can label receptors, block interactions and modulate their function.
Based on our experience in studying structural aspects of cell surface receptors and ligands, we are developing antibody panels that can track receptor presence and modulate activity. We produce truncated antigen constructs for antibody discovery to track receptor activity, ligand binding and structural conformations that affect receptor function. Combining high-throughput yeast display antibody libraries (Kothiwal et al., 2025) with a protein-family approach, we develop specific antibodies that can spatiotemporally follow receptors while a neuron is migrating or forming synaptic connections.
We worked alongside our antibody platform and characterization/validation scientists to develop antibodies for members of families involved in axon guidance and signaling at the synaptic cleft and are now looking toward glial cells as our next frontier. As we expand these protein toolkits, we are engaging scientific communities worldwide to contribute to a fundamental understanding of neuronal differentiation and wiring.

Axon guidance
Axon guidance, or neuronal migration, is the process by which neurons send out axons to reach their targets. It is a critical step in the development of functional neural circuits. This process is regulated by a variety of signaling molecules that either attract or repel growing axons.
IPI has developed antibodies to Glypicans, ROBO/Slits, Netrin/DCCs, Semaphorin/Plexins and members of other families involved in axon guidance. We are currently working to identify additional critical antibodies that enable researchers to detect specific proteins involved in guidance cues and signaling pathways, providing insight into molecular interactions and facilitating the development of targeted therapies for neurodevelopmental disorders.

Synaptic cleft
Synapse formation, or synaptogenesis, is the process by which neurons establish specialized connections with other neurons or target cells, enabling communication within the nervous system. Developing antibody reagents to study neuronal connections is challenging due to the high sequence conservation within receptor families and with other mammalian species, and the dynamic nature of synaptic proteins, which often undergo rapid changes in expression, localization and conformation.
IPI is working on producing highly selective and sensitive antibodies to targets including Neurexin/Neuroligins and Teneurin/Latrophilins. The antibodies may help resolve these challenges and shed light on mechanisms and processes occurring at the synaptic cleft.

Future efforts
We are developing cell surface markers to distinguish glial cell subtypes for phenotyping purposes, using single RNAseq and proteomics data to identify unique cell markers. These will help explore how glial cells support, maintain and protect neuronal activity.
In addition, we are working to develop a standardized set of affinity reagents to enable large-scale exploration of receptor-ligand interactomes and support the development of virtual neuron models to study migration and synapse formation. Additionally, we are creating targeted antibody panels for pathological applications aimed at correlating receptor-ligand phenotypes with disease progression in Alzheimer’s, Parkinson’s and other neurodegenerative conditions.
Key publications
Zhu, S., Jaworski, A., Meijers, R. (2025). Expanding ligand-receptor interaction networks for axon guidance: Structural insights into signal crosstalk and specificity. Curr. Opin. Neurobiol., 92, 102999. https://doi.org/10.1016/j.conb.2025.102999.
Kahn, R. A., Virk, H., Laflamme, C., Houston, D. W., Polinski, N. K., Meijers, R., Levey, A. I., Saper, C. B., Errington, T. M., Turn, R. E., Bandrowski, A., Trimmer, J. S., Rego, M., Freedman, L. P., Ferrara, F., Bradbury, A. R. M., Cable, H., & Longworth, S. (2024). Antibody characterization is critical to enhance reproducibility in biomedical research. eLife, 13, e100211. https://doi.org/10.7554/eLife.100211.
Hao, Y., Yan, J., Fraser, C., Jiang, A., Anuganti, M., Zhang, R., Lloyd, K., Jardine, J., Coppola, J., Meijers, R., Li, J., & Springer, T. A. (2024). Synthetic integrin antibodies discovered by yeast display reveal αV subunit pairing preferences with β subunits. mAbs, 16(1), 2365891. https://doi.org/10.1080/19420862.2024.2365891.
Research news
Axon guidance: Explained
In the late 19th century, neuroscientist Santiago Ramón y Cajal peered through a microscope at the spinal cords of three-day-old…
IPI receives grant from Chan Zuckerberg Initiative to develop next-generation protein tools for neuroscience research
Funding supports creation and open distribution of critical research tools to accelerate neuroscience discoveries Boston, Massachusetts, February 12, 2025 —…
IPI targets neurexin with upcoming antibody suite
Synaptic function involves the interactions of hundreds, maybe thousands, of different proteins. A particular family, called neurexins, has attracted increasing…